Learned, Uncertainty-driven Adaptive Acquisition for Photon-Efficient Multiphoton Microscopy
Cassandra Tong Ye, Jiashu Han, Kunzan Liu, Anastasios Angelopoulos, Linda Griffith, Kristina Monakhova, Sixian You
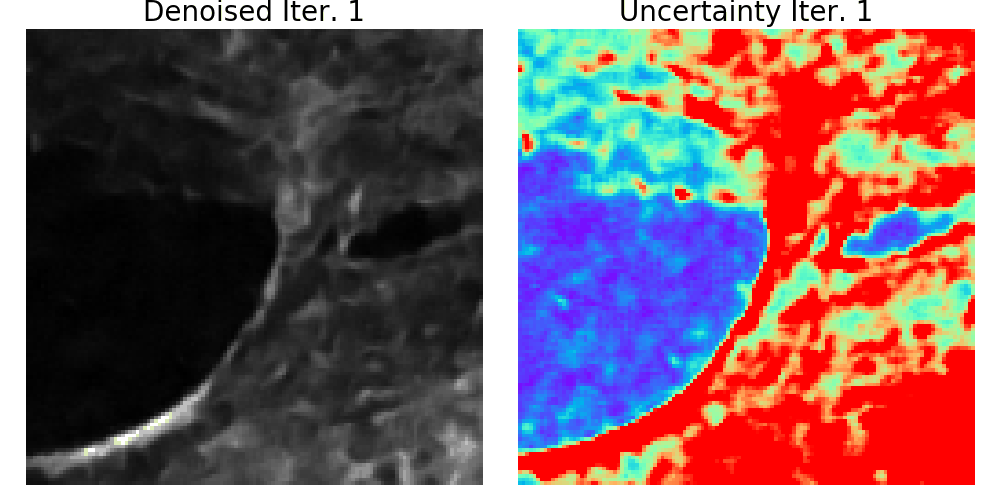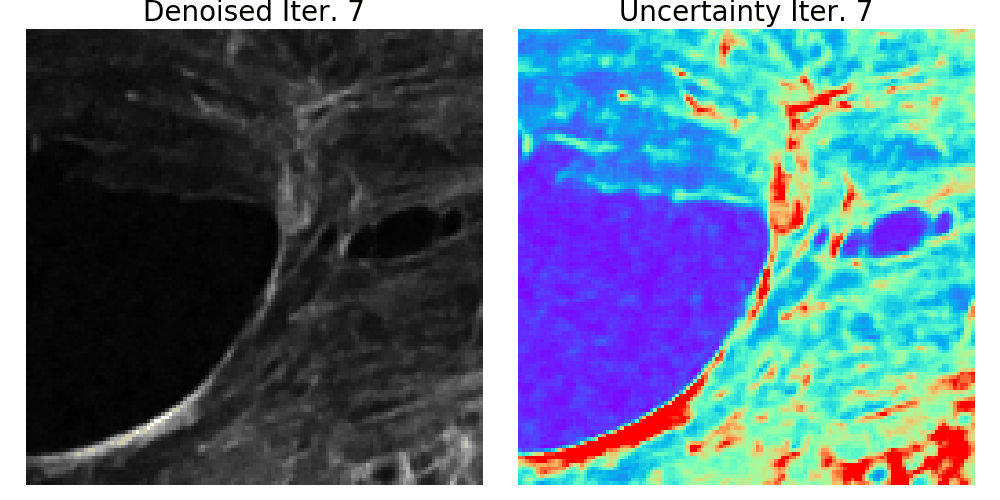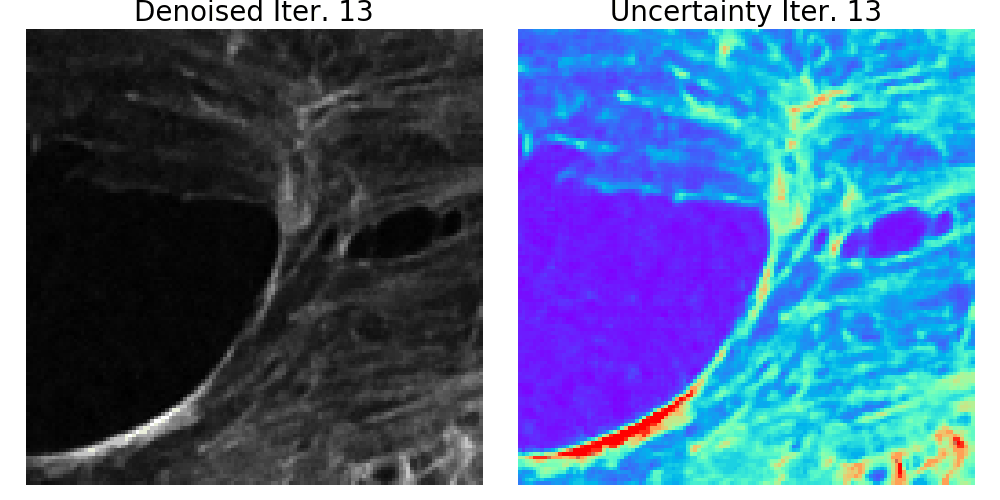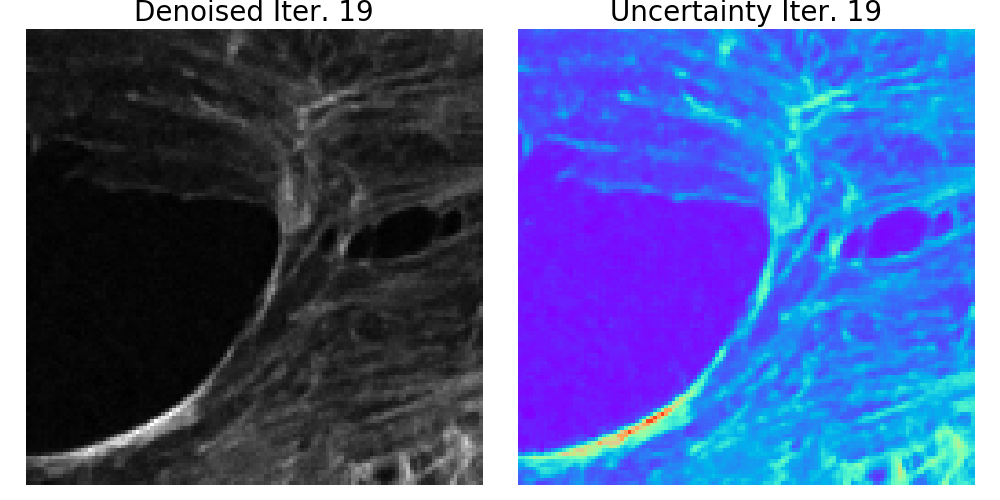
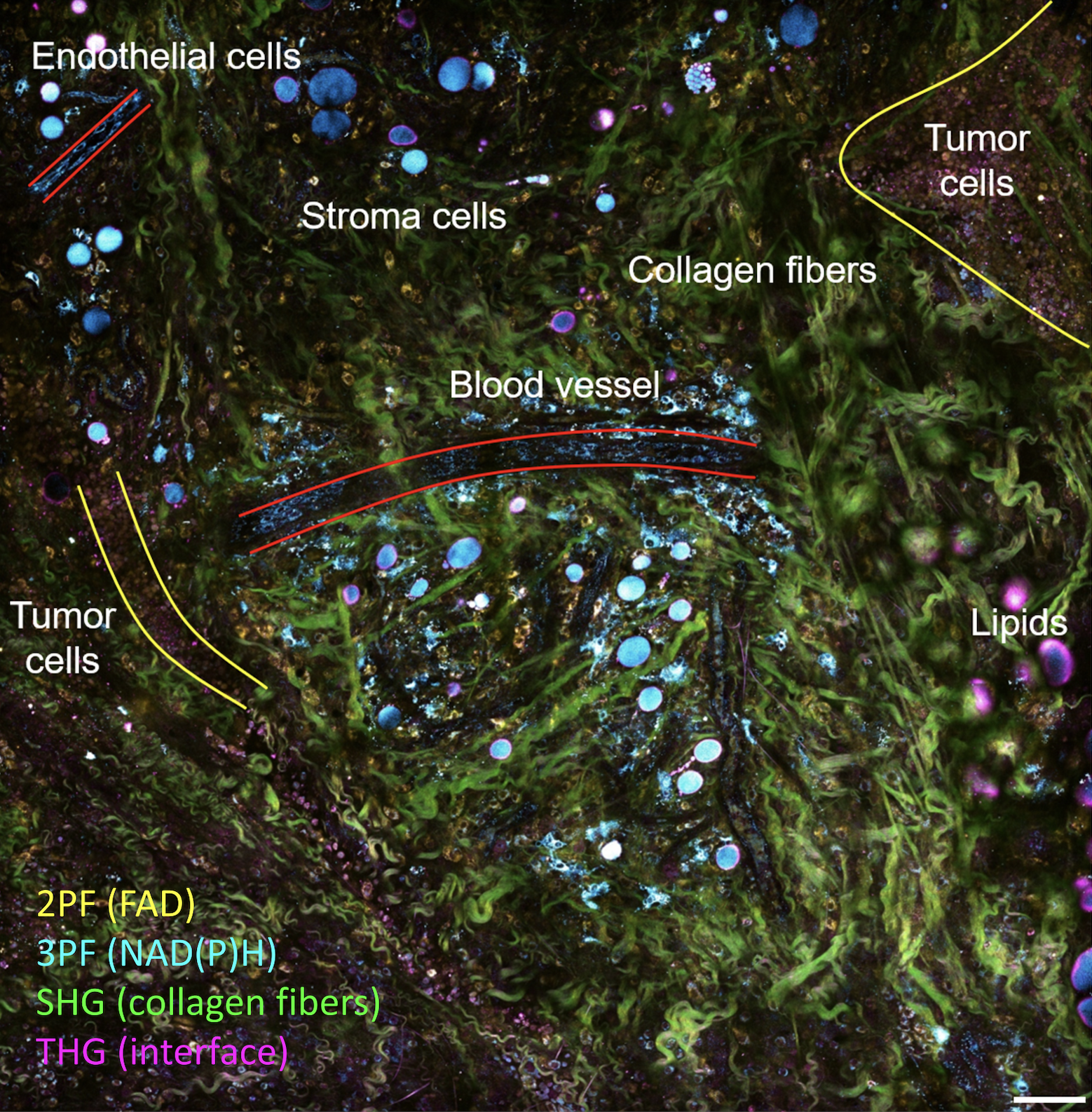
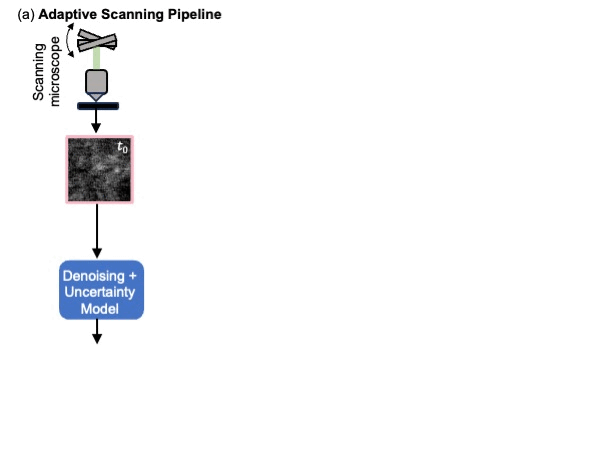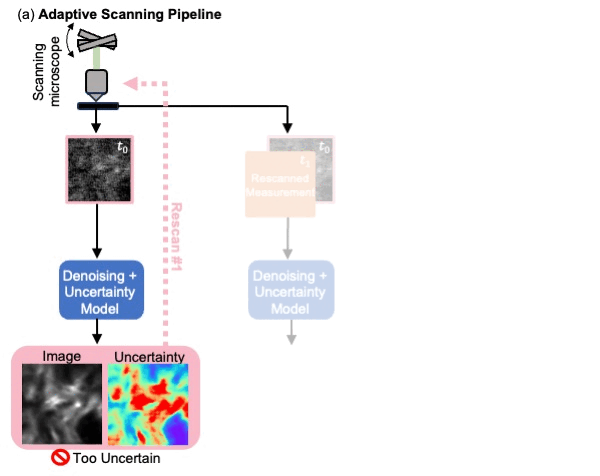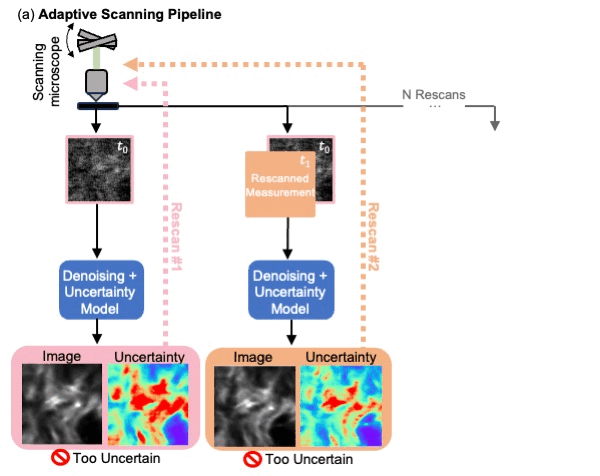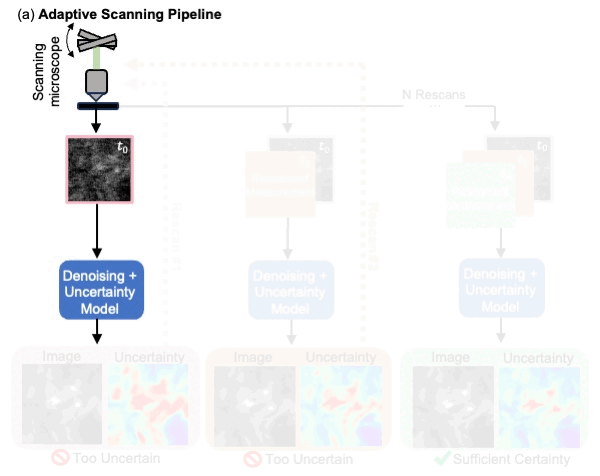
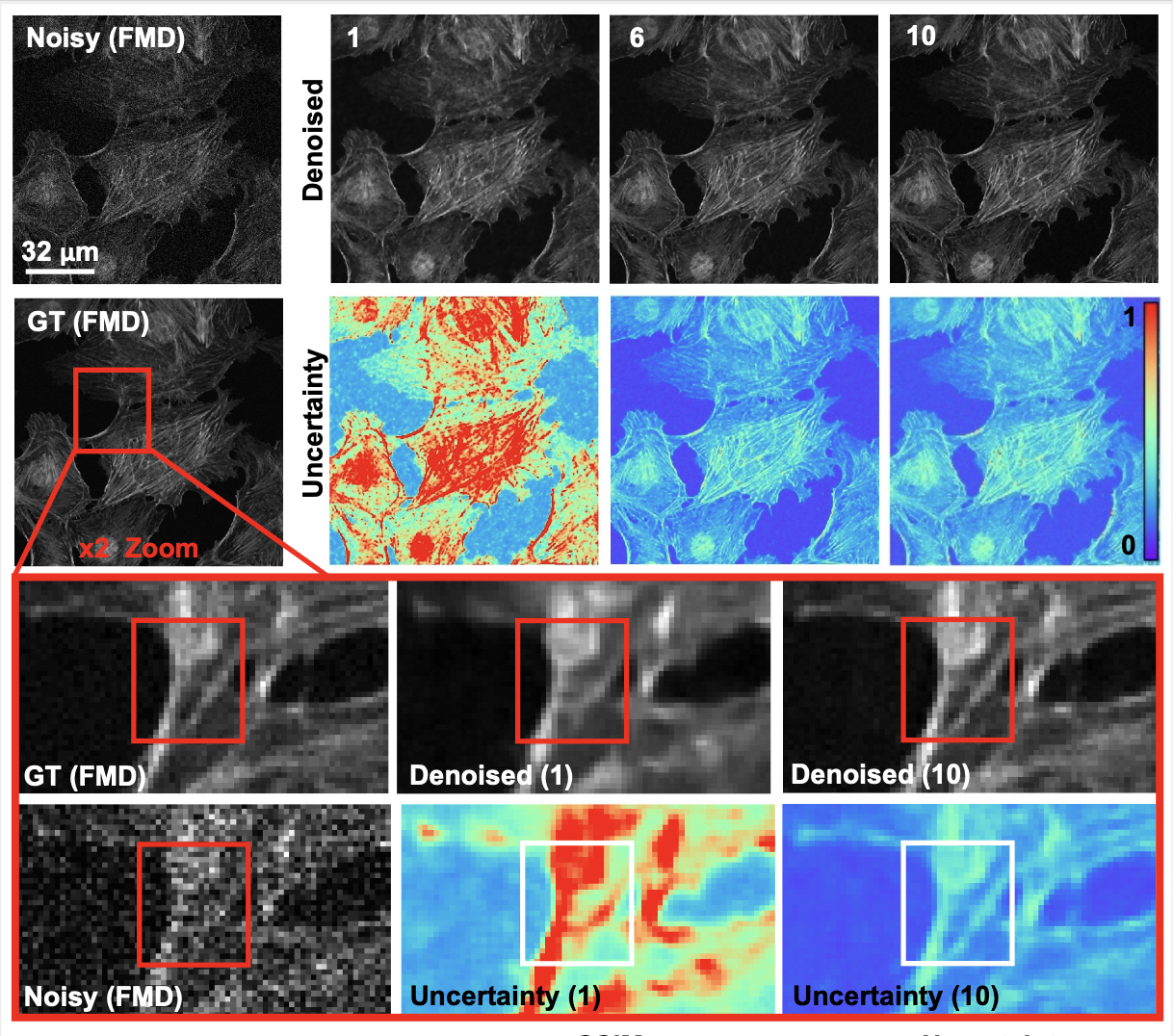
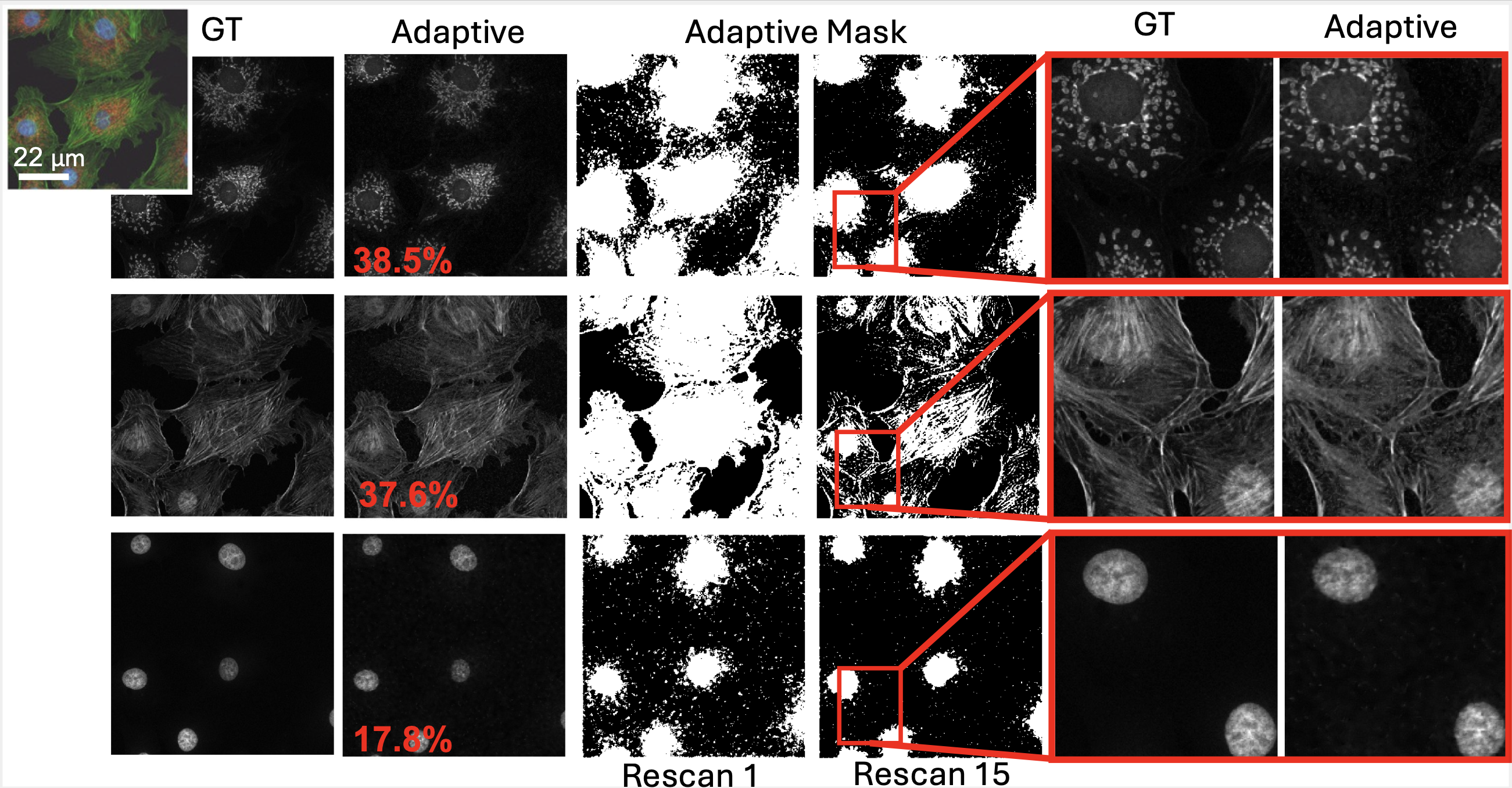
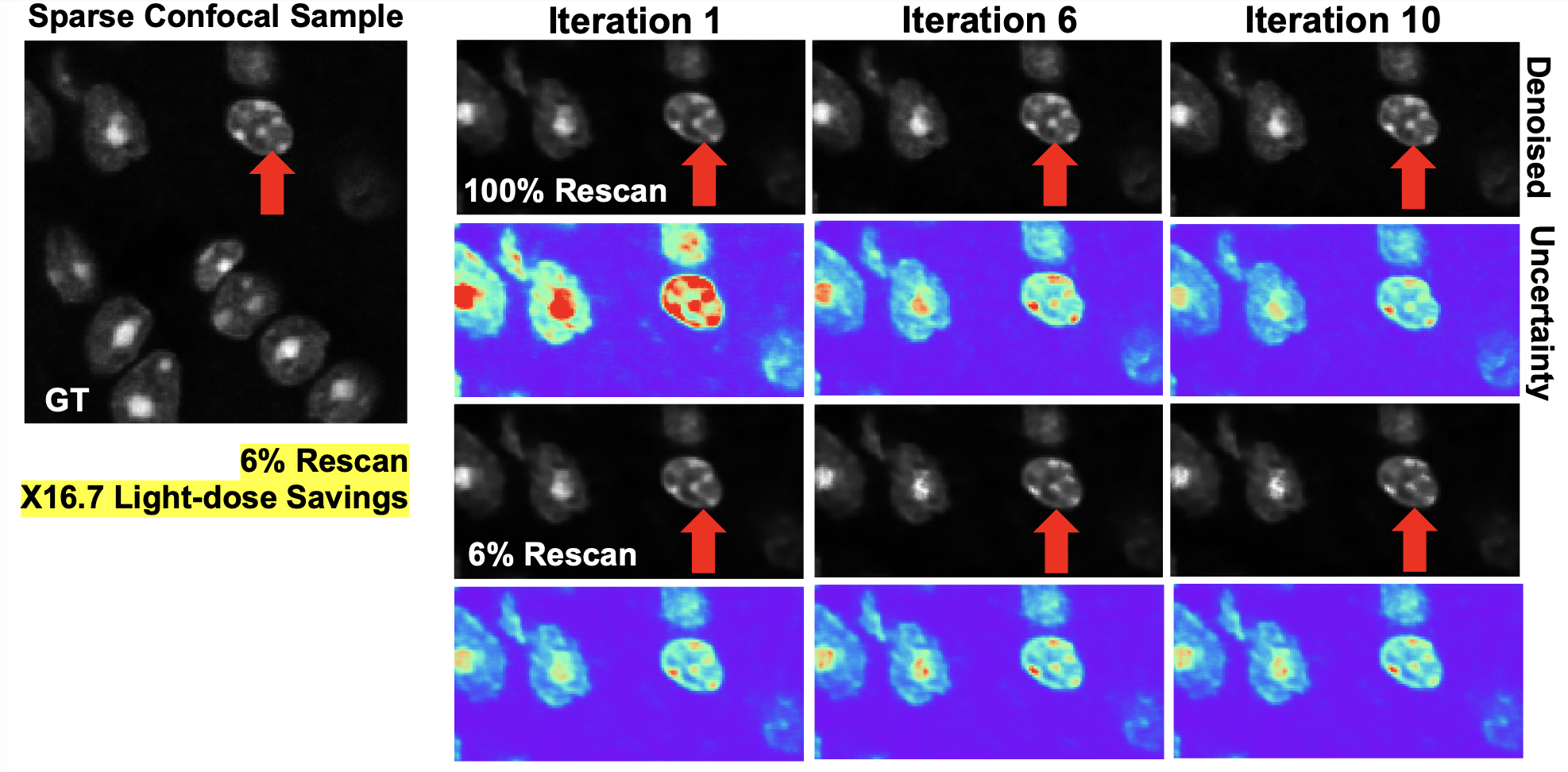
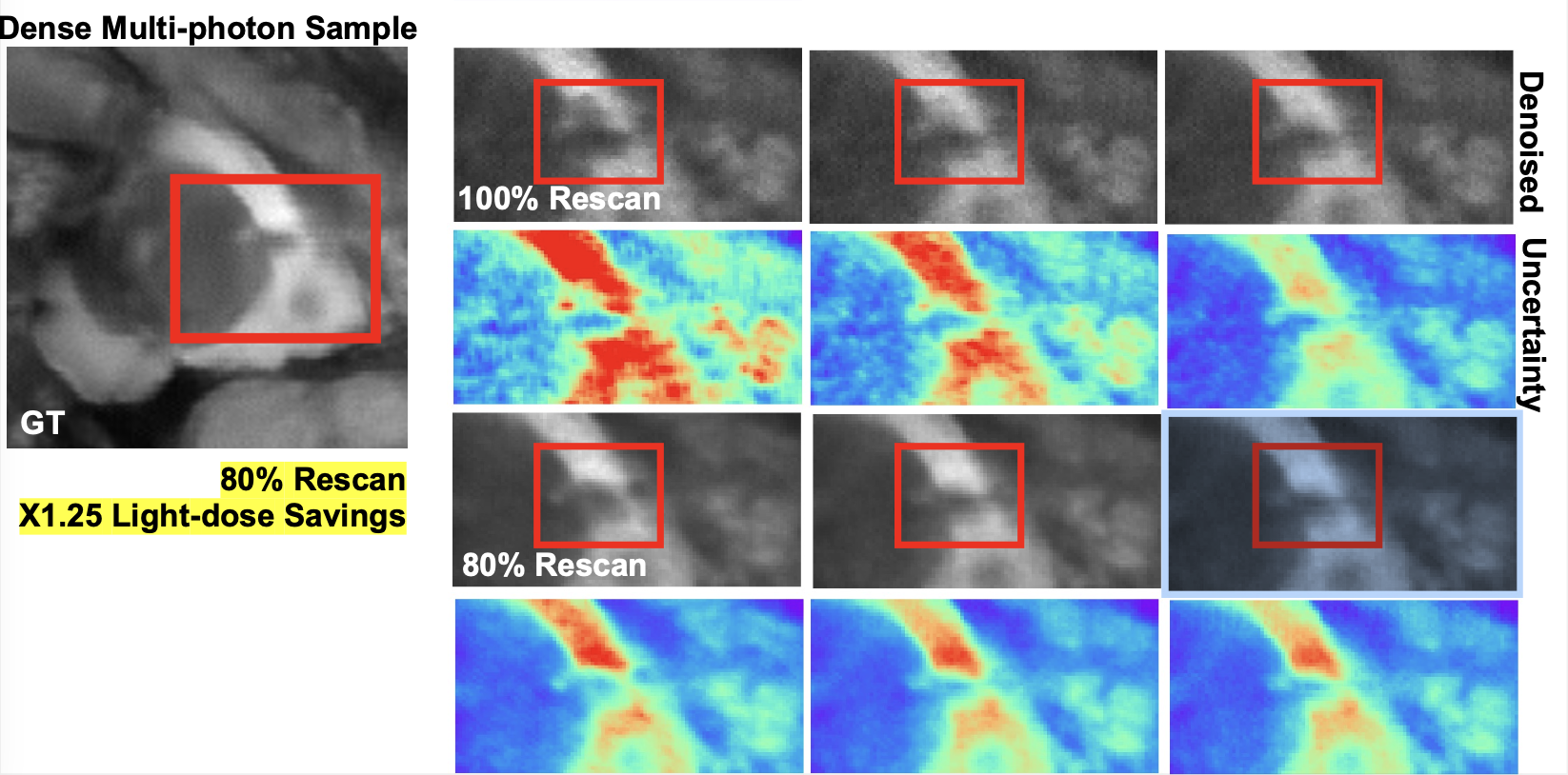
We presented a method to utilize learned, distribution-free uncertainty quantification for multi-image denoising and proposed an adaptive acquisition technique based on the learned uncertainty. In this paper, we demonstrate that our method of uncertainty-driven adaptive acquisition works on experimental confocal, two-photon, and multiphoton microscopy systems, showing a potential 1-16 times decrease in total scanning time and light dose while successfully recovering fine structures. Our method can be adapted for different forms of scanning microscopy without explicit retraining or fine-tuning. Our network trained on FMD data can be used on different data after performing the conformal calibration step using a small calibration dataset. This is one of the advantages of conformal calibration - uncertainty predictions will still hold after calibrating for a different dataset without explicit retraining. All of the statistical guarantees will still hold; however, the size of the uncertainty bounds may increase since the network is not optimized for the data or imaging modality. We discuss this further in our supplement. These speed and total light dose improvements are significant and demonstrate an important step towards faster and gentler scanning microscopy, which will enable the imaging of a new class of interesting samples and lead to new scientific insights and advances.
Furthermore, we demonstrate how deep learning methods for microscopy can be designed to be trustworthy by building in uncertainty quantification to provide error bars for each prediction. Our method successfully identified model hallucinations, which were reduced by taking more measurements or adaptively rescanning the most uncertain regions of the sample. Our method of quantifying uncertainty provides guarantees for the reliability of the prediction. Uncertainty quantification should become standard practice when using deep-learning techniques for scientific and medical imaging to reduce hallucinations and build confidence in image predictions. We believe that the distribution-free learned uncertainty quantification presented here is an attractive path toward this due to its ease of use, fast computational time, and statistical guarantees.

